+603-5131 6160
fleming@cciglobe.com
Medical Device
a.k.a. Conformity Assessment
Medical Device
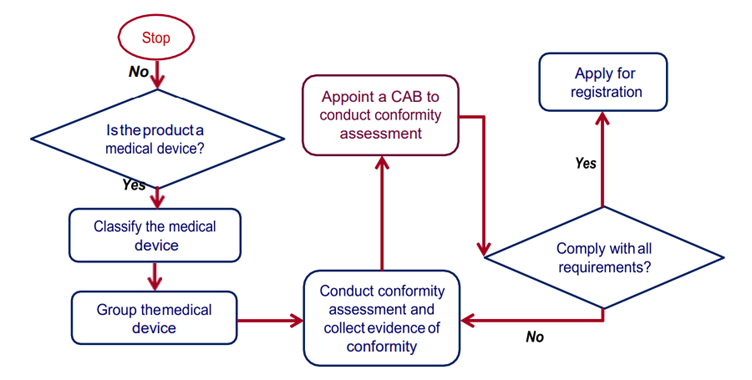
STEPS TO BE TAKEN BY AN APPLICANT BEFORE MAKING AN APPLICATION TO REGISTER A MEDICAL DEVICE UNDER ACT 737.

Elements of Conformity Assessment
- Quality management system (QMS)
- A system for post-market surveillance
- Technical documentation
- A declaration of conformity
Conformity Assessment Type
- By Way of Verification
New Submission
Re-Registration - Full Conformity Assessment
By Way of Verification
- STEP 1: Technical File completion by Establishment for medical devices have Pre- Market Approval from the 6 recognized countries
- STEP 2: Submission to CARE Certification International Sdn Bhd
- STEP 3: Conformity assessment of PMS, QMS, Technical Documentation & Declaration of Conformity
- STEP 4: Establishment License Verification
- STEP 5: Issuance of certificate and report of conformity assessment
Full Conformity Assessment
- STEP 1: Technical File completion by Establishment for medical devices that do not have Pre- Market Approval from the 6 recognized countries
- STEP 2: Submission to CARE Certification International Sdn Bhd
- STEP 3: Conformity assessment of PMS, QMS, Technical Documentation & Declaration of Conformity
- STEP 4: Establishment License Verification
- STEP 5: Issuance of certificate and report of conformity assessment